- Home
- Weddings
- Portraits
- Journal
- Contact
- Download drakorindo runningman episode 409 sub indi
- Mtl rta
- Beautiful anime images
- Foxfi for iphone without jailbreak
- Man of steel superman
- My disney kitchen ps1 price
- Kirby return to dreamland iso ntsc mega
- Controlling live visuals
- Solidworks 2019 break view multiple views
- Katana zero length
- Jewel games free download
- Springfield 1898 krag stock cottage craft
- Restart cherry player
- Rihanna umbrella illuminati
- Jio cinema fire stick
- Antares autotune evo vst rtas v7-0-9- proper-air
- Fallen doll specs
- Winamp for win7 64 bit
- Download black butler season 2 sub indo
- Wp tube plugin pro v2-3 nulled
- Gm service information log in acdelco
- Models mark manson amazonpdf
- Cfx manager 3-1 protocol guide increment
- Terrarium tv app without virus
- Swachhta shapath
- How to get board view files
- Running microsoft project on mac
- Axis camera station s2016
- Free waterlogue app
- Watch sky movies online
- Am i number dyslexic
- Hong kong tourism board 2019
- Papi chulo wrestler
- Ziiosoft total video converter registration code
- Kenwood stereo control amplifier model basic c1
- Movavi video editor 14 plus activation
- Samsung tv
- Antichrist 2009 movie torrent
- Computerized maintenance management system open source
- Seadoo vts position sensor
- Informer snow lyric
- Blue driver scanner
- Hp officejet pro 8720 review
- Movavi video editor 14 full crack
- Sound blaster x fi mb3 free download
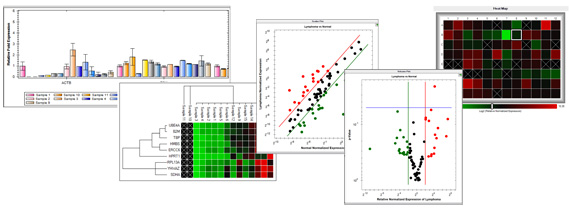
These factors may bias the measured microRNA profiles leading to false-positive discoveries of disease-associated biomarkers. These include the RNA isolation, co-purification of inhibitors of enzymatic reactions, and cellular contamination of the biofluid samples 19– 21. Efforts are ongoing to standardize the blood sampling and processing steps to improve the reproducibility of microRNA analyses 15– 18 ( However, notable sources of variation remain.

Protocols for blood collection, sample processing, storage, RNA isolation, and microRNA quantification often vary across laboratories leading to discordant results 11, 13, 14. One issue is the poor agreement between studies, which may in part be attributed to the lack of standardization 10 and technical difficulties associated with the workflow 11, 12. There are many reasons behind the current unsatisfactory state and their comprehensive discussion is beyond the scope of this article (for reviews see 8, 9). Despite promising advances in the field ( there is still no microRNA test in clinical practice 7, 8. A search for the keywords “microRNA”, “biomarker” and “blood” returns over 5000 hits in the PubMed database (September 2018) with the number of studies increasing every year. After this recognition, a massive wave of research aiming at identifying disease-associated microRNAs followed. The application of the QC panel is demonstrated on the optimization of RNA isolation from biofluids with the miRNeasy Serum/Plasma Advanced Kit (Qiagen).Ĭirculating cell-free microRNAs have emerged in recent years as promising candidates for minimally invasive clinical biomarkers for diagnosis, prognosis, and monitoring of a multitude of human pathologies 1– 6. We provide assay sequences, detailed experimental protocol and guide to data interpretation. The QC panel is a cost-effective way to assess quality of isolated microRNA, degree of inhibition, and erythrocyte contamination to ensure technical soundness of the obtained results. The Two-tailed QC (quality control) panel is based on two sets of synthetic spike-in molecules and three endogenous microRNAs that are quantified with the highly specific Two-tailed RT-qPCR technology. Here, we present a Two-tailed RT-qPCR panel for quality control, monitoring of technical performance, and optimization of microRNA profiling experiments from biofluid samples. Pre-analytical variables including sample collection, RNA isolation, and quantification are sources of bias that may hamper biological interpretation of the results. One of the contributing factors is general lack of agreement between various studies, partly due to the considerable technical challenges accompanying the workflow. Despite substantial efforts invested in the field, the research so far has failed to deliver expected results. Circulating cell-free microRNAs are promising candidates for minimally invasive clinical biomarkers for the diagnosis, prognosis and monitoring of many human diseases.
